 Then the halogens in red (s2 +p5 = 7 valence) compare to the 7 valence electrons present in manganese which is also in red. 1.6 The Periodic Table (a) elements being arranged according to atomic number in the Periodic Table GCSE. Unit 1: THE LANGUAGE OF CHEMISTRY, STRUCTURE OF MATTER AND SIMPLE REACTIONS. Calcium and Zinc both have 2 4s electrons. The Periodic Table can be used to determine whether an element is a metal or non-metal Wales. The same color coding trend is true for the purple. In copper the d orbitals fill up (10 electrons) then the 4s1 fills. The elements copper, gold etc (which are also light blue) also have a single s valence electron. Notice the alkali metals which only have a single s shell valence electron are light blue. 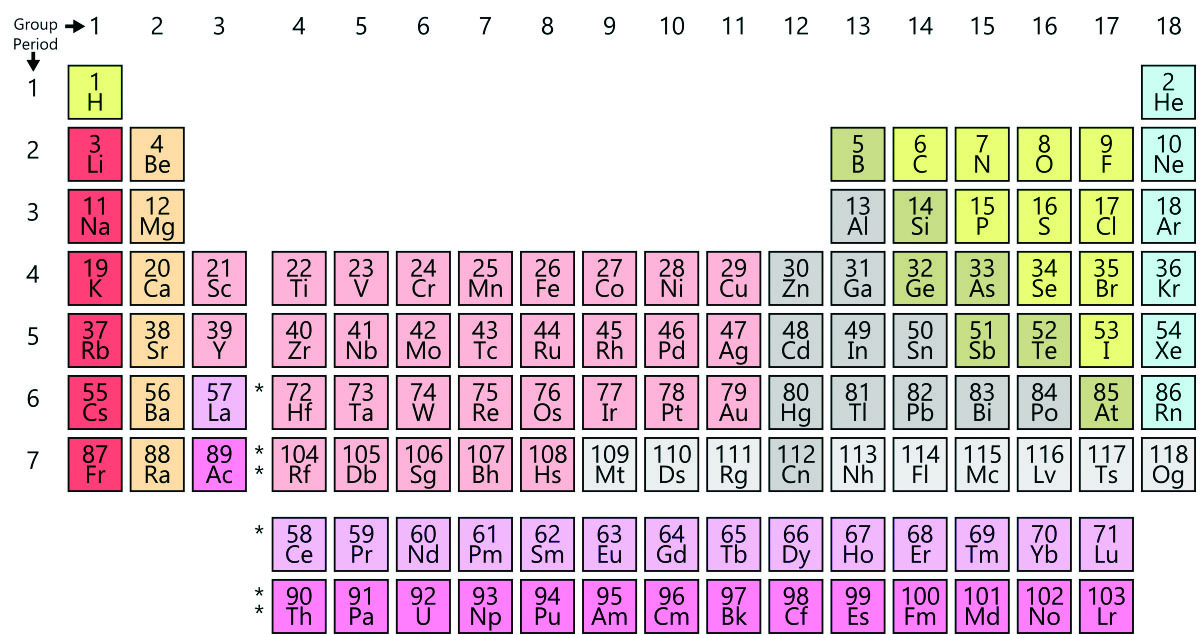
The color coding seems to be really important. That's why the center of the table is expanded. The center of the table has the d-block which can have a total of 18 valence electrons. Do that again until you get to carbon and silicon which are smack in the middle. Move one column towards the center and you gain or lose an electron from the valence shell. On both sides there are full valence shells (noble gases). It looks like it's sorted by the number of valence electrons. Post memes/jokes in /r/chemistrymemes and /r/chemistryjokes. Any such posts will be deleted.Īsk education and jobs questions in the current weekly topic. If you're looking for a more concentrated, advanced discussion of chemistry topics among professionals and grad students, check out /r/Chempros.īefore asking "What chemical is this?" see this chart. Click here for the OSHA chemical data site and here for a multicompany MSDS aggregate search. If you spill/injure yourself contact medical professionals and read the MSDS, do not post to this reddit. 
Yes links to blogs, images, videos, comics, and infographics are okay especially if they are on your personal website. No physorg, sciencedaily, or other press release aggregator spam! If a caption or explanation is included this helps, but please use your discretion.īefore asking about chemical drawing/illustration programs, look at your school's IT/software website and see if they provide an institutional license of ChemDraw (hint: if they have a chemistry department, they will) Likewise, simple pictures of uninteresting and garden variety chemistry-related things are not appreciated. No memes, rage comics, image macros, reaction gifs, or other "zero-content" material. However, academic discussions on pharmaceutical chemistry and the science of explosives are permitted. Rules: Violating a rule will result in a ban.Īsk homework, exam, lab, and other undergraduate-level questions at ChemicalForums otherwise it will be deleted.ĭiscussions on illicit drug synthesis, bomb making, and other illegal activities are not allowed and will lead to a ban. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
